2.3.3 Interpreting conductance in electrolytes
Figure 2.3 shows, how the measurable membrane potential drastically changes in the function of the measuring (clamping) current intensity. At the positions labeled as ”Electrotonic potential” the neuron is in steady state: neither its condenser charges/discharges, nor its ion channels open. When starting from the resting potential, the membrane follows a charge-up function, a saturating exponential. We just note here that the shape of the front side may depend on where the current is introduced. If the microelectrode is inserted directly into the cell, the current causes really a charge-up current. If it is inserted to an axon from an upstream neuron, where no charge carriers are present at the resting potential, the axon must let ions in the tube. This effect also results in a saturating voltage, as experienced by Hodgkin-Huxley [9], see Fig. 3.19. In the two cases the timing constants are similar, but definitely different.
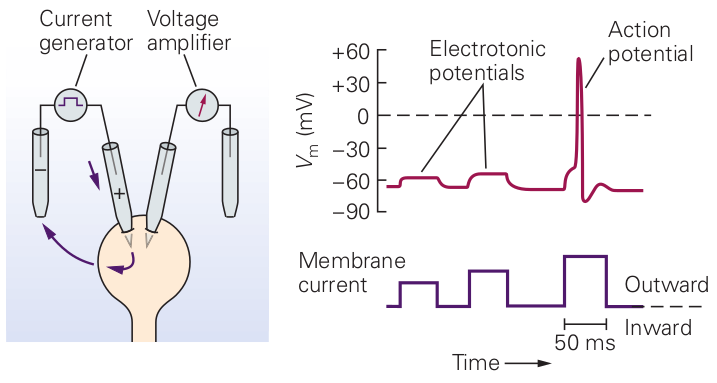
The figure enables one to explain what is misinterpreted in biology in connection with Ohm’s law (and why using a conductance meter in such a case is wrong). Let us divide the membrane current (labeled as ”Membrane current”) with the membrane potential, into regions. In the ”flat” regions (in steady state) we receive the same value and the result can be interpreted as ”conductance”. In those regions the cell behaves ’Ohmic’. However, it is ’non-ohmic’ around the rising and falling edges of the test current, furthermore, if the amplitude of the signal is higher then some threshold value. After receiving the rising edge of the measuring signal, the membrane’s condenser stores part of the charge. If one knows the laws of electricity, fits a saturating exponential to the curve as the membrane potential rises when the condenser charges and the physics is all right. If one believes that the condenser must take instantly the voltage, might say that changes, that is the membrane’s conductance/capacitance changes and has a time constant. Similar explanation can be given, when the falling edge arrives and the condenser discharges. When the current exceeds a threshold value, the ion channels in membrane’s wall open and an extra current (not from the current generator) flows into the membrane. If one believes that the correct value that must be used in deriving conductance is the one that the test generator provides (and the conductance meter does so), divides the test current (instead of the total current) with the measured membrane potential evoked by the total current, uses non-matching value pairs. Non-understanding physics results in claiming non-Ohmic behavior. Neither the conductance nor the capacitance of a cell changes under the effect of electric test signals. Only the measurement is wrong.
Measuring electrolytes
Substances that give ions when dissolved in water are called electrolytes. Certain chemical elements can naturally hold a positive or a negative electrical charge, and they react to their micro- and macro electric environment. A molecule has internal attraction forces that keep its ions in place, and has two charge centers (dipoles). When another dipole or a macroscopic external electric field (which can be of electrical or chemical origin) appears near the molecule, its perturbing effect can affect the relation of the ions to each other. Initially, the two charge centers increase their distance (the molecule polarizes). When that disturbance is strong enough, the ions can entirely separate (the molecule ionizes). The local electric field fluctuates, so the state of the ions is dynamic: they dissociate, free ions recombine. The ions can exist in ionized and polarized states. The state (and behavior) of ions depends on their environment.
A small part (about ) of the molecules dissociates (i.e. the ions leave their counterpart with opposite charge behind) and they move freely in the volume. In other words, the electrolyte liquid can then conduct electricity due to the mobility of the positive and negative ions, which are called cations and anions, respectively. The rest of molecules can be in a more or less polarized state, providing a possibility of producing internal electric field. The macroscopic state of the molecules depends on, for example, how far they are from the boundary of the segment; whether global or local external invasion is applied.
Thermolectric experience shows that, when applying such changes, reaching a steady state is a temporal process, and even the spatial and temporal development of the concentration gradients can be measured as individual processes (the voltage gradient is too fast to measure it). It is also evident from experiments that diffusion is a fast process and that the propagation of the electrostatic field is unimaginably fast ; see our discussion around Eq. (2.24), but it must be process, too. In other words, we have two enormously different interaction speeds. Eq. (2.9) provides only position derivatives. However, Eq.(2.13) and Eq.(2.16) provide the time derivatives for describing the time course of the processes.
In physiology, electrolyte solutions do not surely satisfy the conditions we use for the notions of electricity in physics, see section 2.5. The number of charged objects (the ion concentration) may change in time (even without the presence of biological structures), and a chemical driving force may also move the objects independently from the electrical field. When measuring only the macroscopic electric parameters voltage and current, and especially when measuring current believing we measure directly conductance; in addition, measuring it in a wrong way; (for the details, see section 2.3.3) we attribute the consequences of the injected charge carriers’ low propagation speed to the medium and we describe the phenomenon that ”the conductance changes” in the function of the voltage [112]. (We know that the macroscopical speed of current changes with the clamping speed, see Eq.(2.35) and section 3.9.1), that might change the time difference between the non-matching value pairs, leading to the illusion that the conductance changes.) The measurement must be fixed: the tacit assumptions about notions of electricity must be fulfilled.
Conductance is a “steady-state” notion; see its definition in section 2.3.3 and in section A.3.12 of [2]: ”the input impedance measured after the voltage has reached a steady state following a step change in injected current is defined as input resistance”, or ”the input resistance …obtained by dividing the steady-state voltage change by the current using it” [24]. Using quickly changing (alternating) currents, either sinusoidal or random for measuring conductance, measures some ill-defined current. The experience is resemblant to studying dielectric dispersion in physics. ”Because there is a lag between changes in polarisation and changes in the electric field, the permittivity of the dielectric is a complex function of the frequency of the electric field.” Rearranging charges inside the tested medium needs time. Different polarization types have similar behavior, at much higher frequencies, because of the much shorter distances that rearranging the charges need. In the case of neurons, charges must be rearranged on nearly distances, and ”can no longer follow the oscillations of the electric field” at much lower frequencies; see section 3.10.8. Yes, ”the construction is different from anything we have yet tested in the physical laboratory”.
Physiologists seem to forget the definition and they measure something they think to be ”conductance” in non-steady state. They are ”resetting the clock”, instead of explicitly admitting that the current speed is finite; despite that they measure it to be in the few range. The conductance (per definitionem) does not change; only the (maybe: foreign) charge carriers may need time to deliver the current: we calculate the conductance from non-matching value pairs (or not-steady-state). Wording that biological systems show “non-ohmic behavior” means that they are not metals (they have a charge transfer mechanism differing from the ”free electron cloud”): we abstracted the notion of conductance for metals (or at least steady-state). Physics describes biological operations perfectly; although, it may use ’non-ordinary’ laws (in this case: for slow current). Electric operations are also ohmic in biology, but one has to use the correct (time-aware, i.e., considering the speed of the charged carrier) interaction speed, correct definition and measurement method. Using the Newtonian ’instant interaction’ as the speed of charged ions or the macroscopic speed of their current, is a catastrophic hypothesis and contradicts all our phenomena.
The ohmic behavior means that voltage and current relate to each other, as we learned in college, only when the electrostatic interaction speed is very high (in the mathematical/physical description, the interaction is instant); furthermore, free charge carriers are present in the volume. In biological systems, it is not necessarily the case: the macroscopic speed of ionic current conveying electrostatic interaction is very low, and so they may follow the electrical field propagation apparently with a time shift (if they are improperly distributed, as was early explicitly noticed [9]). As Fig. 3.24 displays, when measuring the secondary entities (instead of a ternary one), everything comes to the right: the voltage and current change using the same time course. Of course, as the are derived form the same primary entity ’charge’. One should measure the voltage instead of assuming the potential appears immediately, even without charge carriers (the locally present individual charges generate the potential). Furthermore, one should not introduce a foreign current into a system (by measuring its conductance or fixing its electric state by adding some feedback current by clamping/patching) when studying the electrical features of that system.
Electrical and chemical invasions
When thermal or electrical invasion happens, the ion’s distribution changes. (Above we assumed an infinitely large volume. Limiting the volume’s size means an asymmetry for the ions in the volume and brings to light unexpected phenomena.) We must also discuss another fallacy that the structured biological objects behave as the metals do under the effect of electrical forces. To derive an abstraction similar to the ones as sciences derive their Laws, we assumed that the ions are tiny charged heavy balls, and they attempt to have a uniformly distributed concentration and potential in the considered space segment. We discuss the cases when an external electrical invasion happens in one segment, when an external chemical invasion happens in one segment, the case when a physical surface mechanically separates the ions in two neighboring segments with different features, when the two separated segments are not symmetrical due to ’Maxwell-demon’-like transmit gates (semipermeable membrane); and when a physical effect concerts the operation of the demons.
The cellular electrodiffusion phenomena are very complex, and it is not a simple task to choose which physical/chemical effects can be omitted so that their omission does not prevent us from explaining physiological phenomena. We discuss mainly the commonly used fundamental omission that the speed of ionic movement cannot play a role in describing neuronal operation.
See also section 2.5.4.