3.9.3 Invasions
Applying a step-like invasion (either concentration of voltage square-wave shaped change)
Concentration square vawe
It has been experimentally investigated the behavior of membrane potential in response to sudden changes of the extracellular concentration of the two permeable ions of the cell. The extracellular concentrations were abruptly changed, as depicted in the left side of Fig. 3.26. According to Eq. (2.9), a negative square wave of the concentration change must provoke a drastic positive potential change, with a time course described by Eq. (2.13). The step-like change results in a step in the voltage, and according to the Stokes formula (see Eq. (2.30)) the ions feel a huge voltage gradient, so they will move with high speed, and an intense current will start. Given that the current means also delivering chemical ions, the membrane tends to find another equilibrium state, corresponding to the newly set concentration, as explained in [2], page 22.
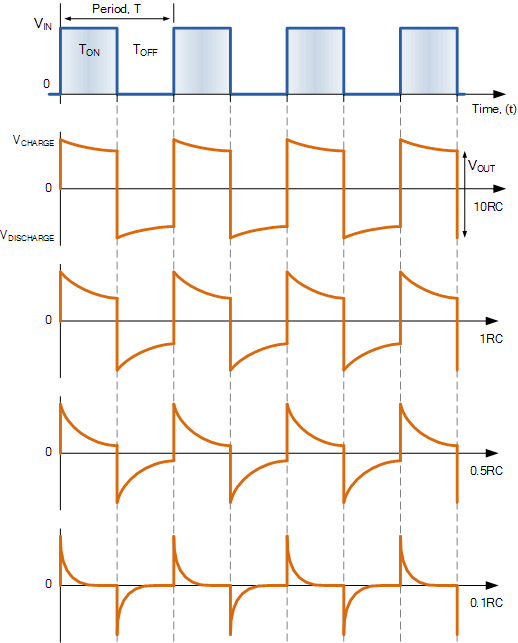
From an electric circuit point of view, the abruptly appearing and disappearing new thermodynamical driving force starts to remove charges and recover charges Essentially, it indirectly discharges and charges the solution. In the right side of the figure, the effect on the output voltage of a differentiator-type electric equivalent circuit is shown under applying a sufficiently long square-ware input voltage change to its input. The negative edge of the concentration change causes indirectly a positive edge in the voltage, and vice versa, so the generated membrane potential should be compared to the reply of the electric circuit to a square-wave input voltage. We can deduce that the serially switched differentiator-type circuit faithfully reproduces the membrane’s electric behavior. From the figure we can conclude that the corresponding time constant can be around .
From the figure we can estimate that the ”half width” of the concentration-provoked potential change is about . (The experiment is not dedicated, we just read back data from a textbook figure.) In Fig. 3.13, we see that the electrically-provoked potential gradient’s half width is about (the lower figure, a relatively short non.rectangle excitation). The experimental value of the ratio of these widths is (it provides the ratio of the corresponding interaction speeds). For the theoretical value see Eq. (2.24).0 If we assume that the propagation speed in the electrolite is , we can conclude potential-assisted speed for the change of concentration (actually, also the potential-assisted speed of current), in line with our other estimations and the published measurements.
Voltage square wave
Similarly, one can apply a voltage square wave to a biological cell, see Fig. 3.27. Compare it to the bottom row of the electrical simulation. From the figure we can conclude that the corresponding time constant can be around .
Notice that the arrival of a square wave evokes
| (3.10) |
ions, in the order of we assumed in section 2.5.7.

Action potential
As we discussed, after the membrane’s potential exceeds its threshold, a membrane-evoked external invasion happens. The suddenly appearing potential change (although it is distributed over its surface) on the membrane is in resemblance with those square-wave excitation functions. Not surprisingly, the amount of charge which evokes an AP is also similar, as we discuss in section 2.5.7.